http://www.hyle.org
Copyright © 2008 by HYLE and Mi Gyung Kim
The ‘Instrumental’ Reality of PhlogistonMi Gyung Kim*
1. IntroductionPhilosophers of science have come a long way in articulating the epistemological and ontological domains for scientific experimentation that are somewhat independent of theoretical representations (Radder 2003). Much like the historical and the sociological studies they draw on, however, their discussions are mostly based on the recent sciences or the experimental practices that employ prepackaged (black-boxed) instruments relying on a stable theoretical framework. Their focus on ‘scientific experimentation’ presupposes a well-established differentiation between theory and experiment, which is not applicable to the laboratory practices that predate the uses of physical instruments, quantitative measurements, and mathematical representations. Chemists were creating artificial realities, however, long before the modern notions of ‘experiment’, ‘theory’, and ‘science’ became available. In other words, chemists could be seen as pioneers in forging the ‘thing knowledge’ and the ‘materialist’ epistemology (Baird 2004). Their knowledge was embodied in chemical substances. Words articulated and justified this material knowledge. Nevertheless, some insights from the newly emerging philosophy of scientific experimentation can be adapted fruitfully to the historical studies of early modern chemistry. For example, Lelas moves away from the traditional notion of scientific theory as "a set of concepts interconnected through laws in the manner of an axiomatic conceptual structure" of mathematics to define it as "a condensed set of instructions of […] how to guide the production of experimental artefacts" (Lelas 1993, pp. 440-442). Affinity tables provide an excellent example of scientific theory in this new definition (Duncan 1962, 1996). If the primary function of modern (physical) instruments consists in the creation of stable phenomena or facts, this function was performed in early modern chemistry by stabilized procedures – distillation and solution methods – which coordinated a set of apparatus and substances in particular order, proportion, and material connection, much like the internal organization of a complex physical instrument. That is, modern ‘apparatus-world complexes’ (Harré 2003) could be seen as the equivalent of the stabilized operations that created artifacts-cum-realities in early modern chemistry. A coordinated instrumentation produces artifacts or bodies that blur the boundary between the natural and the artificial (Kroes 2003). Eighteenth-century chemistry offers an exceptional opportunity for us to examine how chemists created artificial realities and stabilized them as hybrid realities, thanks to their conscious effort to inscribe the esoteric chemical practice in an intelligible language. The project of ‘philosophical chemistry’ required that the new breed of literary chemists translated the material culture of alchemists, apothecaries, and metallurgists into a public language (Donovan 1975, Golinski 1992), which makes it easier for us to discern the "layers of symbolic output" that linked chemical artifacts to chemical theories and philosophies (Lelas 1993, pp. 440-441). In Paris, royal institutions facilitated this process. While the Jardin du roi offered public courses to a mixed audience of apothecaries, physicians, and fashionable men and women of letters, the Academy of Sciences under the direction of Fontenelle pursued a systematic, experimental interrogation of nature amenable to mathematical representation (Fontenelle 1699). In trying to meet the dual demands of teaching and research, French chemical teachers and academicians forged stable material connections, embedded them in a comprehensive theoretical representation of the affinity table, and invented a corpuscular ontology to craft a uniform chemical/philosophical reality. Their efforts to bridge the gap between the artificial/laboratory reality and the supposed natural/philosophical reality can teach us much about the role of instruments in forging a laboratory discipline and the concomitant linguistic and theoretical development (Kim 2003). The changing instrumental/operational identity of fire/phlogiston during the course of the eighteenth century offers a particularly interesting case because of phlogiston’s infamously ‘hypothetical’ status in the Chemical Revolution.[1] In the classic account of this reputedly foundational episode for chemistry, phlogiston was an imaginary substance that eluded detection and positive proof, yet was held responsible for a variety of chemical actions such as combustion and calcination. A metal burned, for example, because it contained phlogiston or the ‘principle of inflammability’, the release of which produced a metallic calx (White 1932). One only had to take a quick look at the weight equation in combustion or calcination process to realize the anomaly that in releasing phlogiston, metals more often than not gained weight. In other words, phlogiston seemingly had a negative weight while it was contained in metals. This logical and empirical impossibility supposedly led to the Chemical Revolution since a substance with a negative weight could not exist in reality (Partington & McKie 1938). J. B. Conant characterized the Chemical Revolution as the development of a ‘superior conceptual scheme’ based on oxygen, which replaced a ‘broad conceptual scheme’ loosely knitted around the hypothetical substance of phlogiston. In this theory-centered account, phlogiston was an ‘erroneous’ theory that hindered scientific progress (Conant 1967). The diversity of opinions regarding phlogiston indicated, according to Partington and McKie, "the last attempts of an incorrect theory to accommodate growing experimental knowledge. Based on an unsound and insecure foundation, such a theory […] at last becomes so top-heavy that it crashes by mere instability of bulk" (Partington & McKie 1937, p. 361). The inductivist and the falsificationist accounts of the Chemical Revolution have been subjected to various modifications in the post-Kuhnian era. In characterizing the Chemical Revolution as ‘a particularly famous example’ of a paradigm change or a revolution in science, Kuhn’s model of Gestalt switch (Kuhn 1962) rendered phlogiston chemistry more rational and provided a fresh impetus for a detailed historical investigation of Lavoisier’s path to the Chemical Revolution that was underway.[2] In subsequent philosophical modeling, phlogiston and oxygen became competing theoretical frameworks that allowed chemists to make sense of an ensemble of empirical data (McCann 1978). Further elaborations of Kuhn’s model took into account the historical investigation then available to make the lingering opposition to Lavoisier’s revolution more reasonable. Alan Musgrave employed Lakatos’ notion of research programs to rationalize phlogistonists’ multiple ontologies and their lingering adherence to the theory despite obvious anomalies. Only a ‘superior’ research program could overthrow the existing program and initiate a revolution in science (Musgrave 1976). Howard Margolis further characterized the Chemical Revolution not as a rational theory choice based on the logical gap in experimental data, but as a psychological process governed by the ‘habits of mind’. Paradigm shifts require that the historical actors overcome the barriers long cultivated by existing practices, institutions, and cultures (Margolis 1993). While these philosophical models of the Chemical Revolution accord a level of rationality to phlogistonists, they still revolve around the ultimate ‘overthrow’ of the phlogiston theory (Thagard 1990, Pyle 2000). In other words, they ignore the historical fact that phlogiston did not provide a comprehensive theoretical framework for pre-Lavoisian chemistry. It was a relatively ordinary substance involved in a multitude of chemical actions (Toulmin 1957, Perrin 1992, Shelton 1995). Despite the increasing sophistication in modeling the Chemical Revolution, then, philosophers have not yet integrated recent historical investigations on pre-Lavoisian chemistry to reassess the nature of this episode. Most important in this regard is F. L. Holmes’ groundbreaking work that identified the main contour of eighteenth-century French chemistry in the investigation of ‘salts’ (Holmes 1989).[3] He has convincingly shown that pre-Lavoisian chemistry possessed well-defined objects of investigation and investigative paths. I have argued elsewhere that the shift in the main method of chemical analysis, from the distillation of natural bodies to the displacement of stable substances in solution, entailed a change in the ‘model system’ that produced the ‘objects’ of chemical inquiry. The notion of chemical composition changed accordingly from an arrangement of property-bearing principles to a dualistic one determined by selective chemical action, which was represented in the affinity table (Kim 2006).[4] French chemists worked within a well-defined research field of salts (akin to Kuhn’s notion of normal science) equipped with a clear, methodical representation of their operational knowledge. In the affinity table, phlogiston became a substance well characterized through the operations involving various salts, rather than a free-standing theoretical construct that guided chemical research until the advent of pneumatic chemistry made it a problematic entity. In other words, we can no longer assume that phlogiston was an "irresistible intuition" or a central concept that lent "coherence to a variety of superficially unrelated phenomena" for its defenders (Margolis 1993, p. 59, 49), much less that chemistry was mere cookery before Lavoisier rationalized its haphazard practices. If we acknowledge the importance of salts in
establishing main theory domains and theoretical representations of
eighteenth-century French chemistry, we need a radically different
understanding of why phlogiston became the rhetorical focus of
Lavoisier’s campaign to reform chemistry. In order to redress the
central position phlogiston still occupies in the narrative of the
Chemical Revolution, we need a more historically sensitive
understanding of its evolving identity in chemical practice, theory,
and philosophy. As a step in this direction, I argue in this paper that
phlogiston was an integral part of chemists’ laboratory reality
stabilized by their techniques of analysis and synthesis, theoretical
representations, and philosophical commitments. At least in the early
eighteenth century, it was as ‘real’ as any other chemical substance,
even if chemists could not exactly put it in a bottle and label it. To
appreciate this ‘instrumental’ reality of phlogiston (cf. Ihde
1991), we need to understand its operational identity in
distillation methods as ‘an oily matter’, its theoretical identity
represented in the affinity table as a salt ‘principle’, and its philosophical
identity
in corpuscular ontology as ‘the matter of light or fire’. The
philosophical identity of fire/phlogiston rested in part on its
instrumental association with the burning glass and in part on the
‘speculative’ ontology derived from a prior commitment to the ‘true’,
uniform representation of nature.[5]
These variegated identities forged in different
operational/instrumental, theoretical, and philosophical contexts were
intricately interwoven in the didactic discourse to stabilize
phlogiston as a chemical substance. The evolution and maintenance of
fire/phlogiston as a stable substance depended on chemists’
inventiveness in forging material connections, theoretical
representations, and philosophical legitimacy. Changing operational
(instrumental) methods could therefore cause a rupture in the existing
nexus of reality and meaning. Herman Boerhaave’s thermometric
conception of fire stipulated weightless particles of fire, which
undermined its capacity to function as a chemical substance or
phlogiston as well as its instrumental association with the burning
glass. A new physical instrument demanded a new ontology. The efforts
to accommodate this radical intervention in chemical theory resulted in
a rather inchoate set of definitions for phlogiston, which made it
vulnerable to Lavoisier’s attacks. 2. The Sulphur PrincipleThe history of phlogiston in France began at the turn of the eighteenth century with Paris Academicians and their quest for the ‘sulphur principle’ led by Wilhelm Homberg (1652-1715), who played a crucial role in forging a theoretical discourse of chemistry that chimed with the newly popular Cartesian philosophy and Fontenelle’s vision for a unified science based on intelligible principles and the interrogation of nature (Fontenelle 1699, 1715). In order to bridge the gap between chemists’ laboratory reality and philosophers’ speculative ontology, Homberg deployed a new instrument of analysis (the burning glass) and tried to correlate its products with those of existing chemical methods (distillation and solution methods). His intervention to constitute a uniform chemical/philosophical reality left important residues in chemical theory that persisted throughout the eighteenth century. To trace theses residues, we need to begin with his analysis of metals (Kim 2000). The guiding thread in Homberg’s quest for a uniform chemical/philosophical reality was the ‘sulphur principle’ – the active principle in all bodies, a definition somewhat reminiscent of the alchemical quest (Principe 2001). Although he deferred initially to chemists’ understanding of this principle as the final point of analysis – the ‘oily or fatty matter’ extracted from various vegetable, animal, and mineral bodies through distillation – he began to explore the possibility of further reducing this ‘chemical principle’ to its ultimate corpuscular identity when a Tschirnhaus burning glass was made available to him (Homberg 1702a). He believed that this powerful furnace would allow him to command pure fire, nature’s most effective solvent, to reveal the ‘true sulphur principle’ that composed various sulphur principles chemists dealt with in their distillation or solution analysis. In his hierarchical understanding of composition, natural bodies contained various sulphur principles that chemists could extract through chemical analysis (metallic, bituminous, and vegetable sulphur or inflammable oil), but all of these sulphur principles would contain the ‘true sulphur principle’ – a completely pure matter that escaped chemists’ analysis and senses, yet conferred activity on all bodies. He identified this elusive principle as the ‘matter of light’ (Homberg 1705, 1706). In the following diagram, [N] refers to the domain of nature, [A] to the domain of reality constructed by actual chemical analysis and [P] to the domain of reality justified by chemical philosophy and projected analysis.
Homberg’s ‘true sulphur principle’ was not a weightless fluid, much less a substance of negative weight, but its reality and weight depended on the burning glass. Robert Boyle, Samuel Du Clos, and other seventeenth-century investigators had all assumed, based on the burning mirror experiment, that fire was a substance that participated in chemical actions and augmented the weight of the affected bodies. To establish the involvement of pure fire in chemical action as an ‘incontestable fact’, therefore, Homberg repeated Du Clos’ 1667 experiment on the regulus of antimony with the Tschirnhaus burning glass, more powerful and convenient than the old burning mirror, and the regulus of Mars (iron).[6] Since no other matter touched the regulus during the calcination process, he attributed the weight gain in the calx to the solar rays or ‘the matter of light’, which therefore had to be the sole active principle in the universe or the sulphur principle. To prove the universal applicability of his instrument and thereby to establish his hierarchical theory of composition, however, Homberg had to correlate the products of the burning glass analysis with those of chemical analysis (Homberg 1708). The elusive ‘true sulphur principle’ could acquire its material justification only through a set of more mundane chemical analysis and theorization thereupon. Forging material connections between the different methods of analysis would have been a tedious endeavor, as we shall see in Geoffroy’s efforts. Instead, Homberg built an elaborate speculative scheme using the matter of light as the sole active principle in the universe. In his scheme, the matter of light could combine with another principle to produce four different kinds of sulphur – metallic, bituminous, vegetable, and animal sulphurs – as we have seen in the previous diagram. Any of these sulphurs could then combine with a pure acid to produce an acid spirit, which in turn could combine with an alkali to constitute a middle salt. In principle, therefore, chemists should be able to transform one kind of sulphur to another, an acid to a different acid and a middle salt to another salt. Analysis does not simply reduce the phenomena to their elementary simplicity, but also it allows scientists to project new phenomena "unobserved, or still to come, or to be brought about" (Lelas 1993, p. 433).
The corpuscular imagery provided an additional rationale to render esoteric chemical processes intelligible to the literary public. In Homberg’s scheme, metallic sulphur was nothing but the matter of light that penetrated and attached itself to the globules of mercury with its natural glue. The proportion and the strength of this binding, coupled with the presence of other matters, determined various kinds of metals. The burning glass supposedly offered Homberg, then, a privileged access to the corpuscular level of composition, much as our instruments of particle physics are meant to reveal the innermost constitution of matter. He hoped that it would perform an ultimate dissolution beyond the analytic efficacy of ordinary solution methods (using acids and alkalis) and solve the dilemma of the composition-altering distillation method. It could thereby provide a common instrumental foundation for chemical analysis. By weaving together different analytic methods, a classificatory scheme, and a corpuscular ontology, Homberg attempted to provide a consistent chemical/philosophical reality for the sulphur principle, which acquired in his system a discursive reality that represented nature to both philosophers and chemists. The credibility of Homberg’s system rested in part, however, on the trust the Tschirnhaus burning glass could command among chemists. The instrument was not accessible to most chemists, nor was it applicable to all bodies.[7] Chemists who learned their art in the pharmaceutical trade were not easily persuaded by Homberg’s speculative scheme centered on an elusive substance. The depth of this divide between a natural philosopher/chemist and an apothecary/chemist becomes evident in the divergent approach to the composition of metals by Etienne-Francois Geoffroy (1672-1731) whose investigation established the classic operations involving phlogiston – the calcination and the reduction of metals (Kim 2003, pp. 96-103). An heir to a pharmaceutical dynasty, Geoffroy had an extensive training in the trade before he became a member of the Academy (Fontenelle 1731, Planchon 1898). While Homberg tried to streamline complex chemical operations with the use of a single physical instrument in support of his prior commitment to the uniform composition of nature, Geoffroy sought to stabilize concrete operations and bodies and thereby to establish material connections among them. Soon after Homberg announced the decomposition of common (mineral) sulphur into three main components, therefore, Geoffroy ‘proved’ it by recomposing common sulphur as well as iron from their three decomposition products – a vitriolic salt, the sulphur principle and an earth. This ‘philosophical proof’ indicated that common sulphur and iron differed only in the earth they contained, sharing two other common constituents (Geoffroy 1704). Geoffroy pursued Homberg’s philosophical project – the uniform composition of nature – through the material operations of analysis and synthesis in establishing the general composition of metals. Perfect metals did not cooperate. Although the vitrification of gold and silver with the burning glass proved that they contained a sulphur principle, he was not able to revive their metallic nature by adding the sulphur principle contained in charcoal. Imperfect metals proved more cooperative. By 1709, he could offer experiments to support his thesis that a sulphur could restore metallicity to imperfect metals such as iron, copper, tin and lead. With the burning glass, he could vitrify these metals in a vessel that did not supply the sulphur principle and then restore their metallicity by placing the vitrified metals on charcoal and melting them. This reversible operation proved to Geoffroy’s satisfaction that charcoal supplied the single principle the vitrified metal needed to regain their metallicity. He thus concluded that each of the four imperfect metals was composed of an earth susceptible of vitrification and a sulphur (Geoffroy 1709). This reversible process became a stable component of phlogiston’s operational identity in France, although it was later attributed to Stahl. The ‘sulphur principle’ invariably meant for Geoffroy, however, a concrete oily substance separated out in chemical analysis, rather than Homberg’s ‘true sulphur principle’ or the matter of light. Geoffroy also rejected the idea that mercury – long considered a common constituent of all metals and the principle in Homberg’s scheme that supposedly produced metallic sulphur by combining with the matter of light – could enter into any metal. Sulphur, acid, and earth sufficed to compose a metal. Their relative doses, strengths of union, and manners of uniting accounted for all differences among metals. If all metals had a composition similar to that of iron, sharing their sulphur principle with common sulphur, one could effect the transformation of metals into common sulphur to prove that the sulphur principle was the sole active principle in the universe. As the solution analysis became more entrenched in chemical practice, Louis Lemery strengthened the role of the sulphur principle as the universal solvent by conjuring up a detailed corpuscular ontology of the ‘matter of fire’ (Lemery 1709). By establishing a strong analogy between the functions of water and fire, he conceptualized the previously disparate chemical actions involving them as displacement reactions that could be theorized in a similar manner (Kim 2003, pp. 111-132). As the ‘first & most powerful solvent of earthly bodies’, the matter of fire penetrated deeply and disunited ‘the essential substances’ much more perfectly than any other known agent. Often, however, it failed to dissolve solid bodies and became imprisoned in them, as in the calcinations of antimony, lead, tin, and mercury with the burning glass, which markedly increased the weight of the body. This ‘fixed fire’ conserved its nature, however, much like the water fixed in many different bodies. Lemery clearly distinguished the ‘fixed fire’, insensible to the touch, from the ‘free fire’ that could pass between bodies in the form of heat. Accordingly, there existed two kinds of pores: those sufficiently large to give a free passage to the matter of fire all the time and those that allowed the passage only when dilated by heat. Lemery’s distinction between the ‘fixed fire’ and the ‘free fire’ thus accommodated the phenomena of heat in conceptualizing chemical processes and composition. More significantly, Lemery articulated the
displacement reactions of metals in acid solutions that produced the
‘true’ precipitates as the model system in chemical analysis. Mercury
precipitates offered a particularly interesting case because, unlike
other metallic precipitates, they acquired distinct colors depending on
the absorbent salt used to precipitate them (Lemery 1711). Not only did
the color change with the salt used, but also its intensity depended on
the relative quantities of the acid and the absorbent salt. Lemery
attributed these color variations to the quantity of the ‘parts of
fire’ contained in the precipitate. This correlation made sense to him
since the alkaline property of the absorbent salt depended on the fire
it had previously accepted. In acid solution, the alkaline salt would
exchange these parts of fire for the acid parts, making the parts of
fire available to mercury. Lemery’s attribution of color to the parts
of fire probably made it easier for the later generations of French
chemists to accept Stahl’s designation of phlogiston as the principle
of color (Lemery 1712). Although Lemery did not design new experiments,
his careful reasoning patched together various operational
(instrumental), theoretical, and philosophical identities of the
sulphur principle to form a more or less stable discursive identity
that provided a clear precedent for Boerhaave’s discussion of fire in
his popular Elements of Chemistry. Commonly referred to as the Traité
du feu, it became an integral part of French discourse on heat,
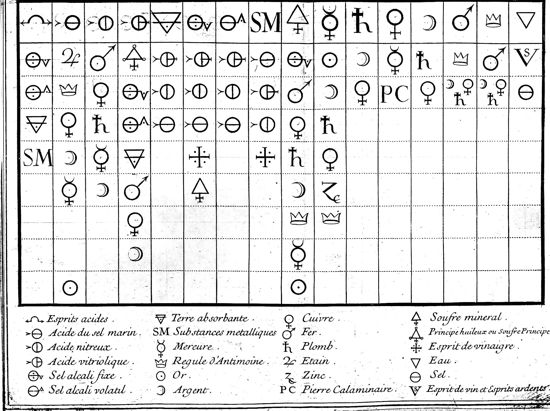
fire, and phlogiston for the remainder of the eighteenth century. 3. PhlogistonThe ‘sulphur principle’ in the early decades of the eighteenth century carried at least two distinct meanings derived from different kinds of fire analysis – ordinary chemical analysis and the burning glass analysis. The operational identity of the sulphur principle as an oily substance derived from the distillation analysis, while the philosophical or speculative identity of the ‘true sulphur principle’ as the ‘matter of light’ was based on the burning glass analysis and subsequent interpretations. These two identities were mediated by its theoretical identity as a ‘principle’ of acids and salts, which soon acquired a secure place in the systematic representation of chemical operations in solution. In 1718, Geoffroy presented to the Academy his Table des différents rapports observés entre différentes substances, which summarized the basic operations of solution chemistry in a tabular form so that "a beginner will form in a short time a just idea of the rapports different substances have with one another & chemists will find an easy method for discovering what happens in several of their operations difficult to unravel & what has to result from the mixtures they make from different mixed bodies" (Geoffroy 1718, p. 203). In its pedagogical and predictive functions, Geoffroy’s Table des rapports (later known as the affinity table, see Figure 1) served as an effective theoretical representation of chemical operations in lieu of the corpuscular ontology. Embedded in this table, the sulphur principle (later changed to phlogiston) acquired a stable theoretical identity that would sustain its place in the evolving chemistry of salts. To understand the staying power of phlogiston in pre-Lavoisian chemistry, therefore, we must unpack this operational/theoretical identity of the sulphur principle as it was presented in the affinity table. As is well known, Geoffroy’s Table des rapports represented basic operations involving acids and alkalis. The top row listed sixteen substances chemists commonly employed in solution chemistry. Below each of them were listed the substances that reacted with the top substance in the decreasing order of their ‘rapports’ with it. For example, the first column was headed by acid spirits, which was followed by fixed alkali salt, volatile salt, absorbent earth, and metallic substances. This column represented the chemical experience that fixed alkali salt combined most strongly with acid spirits and would displace all other substances listed below it from their existing combinations with acid spirits. Likewise, volatile alkali salt would displace absorbent earth and metallic substances from their combinations with acid spirits. It would not, however, displace fixed alkali from its combination with an acid spirit. The left half of the table, columns one to eight, summarized the displacement reactions of acids and alkalis that formed middle salts. The right half of the table, columns nine to fifteen, summarized the well-known operations in metallic chemistry that formed alloys (Klein 1995). Both halves of the table follow the same principle of displacement according to fixed rapports, if one understands the first half as reactions in the solvent of water and the second half as reactions in the solvent of fire. Although Geoffroy does not cite Louis Lemery’s contribution, conceptualizing fire as a solvent was a necessary move to provide a uniform theoretical understanding of the model system in solution chemistry.
Figure 1: Geoffroy’s Table des rapports (from Geoffroy 1718). Geoffroy included the ‘oily or sulphur principle’ in the fourth column of his table and soon identified it with ‘phlogiston’ in discussing a chemical puzzle earlier proposed by Stahl: "When one has saturated & crystallized vitriolic acid with salt of tartar, find a means of separating this acid from the fixed salt in a moment of time & in the palm of a hand." (Geoffroy 1720) This seemed a rather difficult puzzle, Geoffroy mused, since salt of tartar was the strongest alkali and should have ‘the most intimate rapport’ with vitriolic acid, which was the strongest acid. It was quite unlikely that any other acid or alkali could break up their union, as Stahl himself had acknowledged in his Traite de Zymotechnia or De formentatione. By carefully sorting through Stahl’s works, Geoffroy answered that the only substance that could displace salt of tartar from its union with vitriolic acid was the "oily principle, or as M. Stahl calls it, the phlogistic principle, the inflammable principle, or the principle of inflammability" (ibid.). He devised a couple of processes to prove the involvement of this principle (Kim 2003, pp. 146-151). In one of these processes, Geoffroy melted vitriolated tartar (salt of tartar saturated by vitriolic acid) on a crucible with some inflammable matter to produce a reddish matter similar to hepar sulphuris (a mixture of salt of tartar and sulphur). By dissolving this mixture in a sufficient quantity of water, filtering it, and pouring it over some distilled vinegar or other weak acid spirit, he obtained sulphur.
Geoffroy interpreted that in this operation the oily principle contained in the inflammable matter, rarefied and put in motion by the element of fire, insinuated itself between the two salts that made up vitriolated tartar. Since it had more rapport with vitriolic acid than with the alkali salt, it united with the former quite tightly and consequently detached itself from the latter. The ‘compound’ of common mineral sulphur resulted from the union of the oily principle and vitriolic acid. The salt of tartar, abandoning vitriolic acid, remained united with this new sulphur in the form of hepar sulphuris. To facilitate their separation, one had to pour some acid for which the alkali salt had a much more considerable rapport. Geoffroy identified his ‘oily or sulphurous
principle’ with Stahl’s ‘phlogiston’ or ‘principle of inflammability’,
by following every minute detail of concrete operations that supposedly
involved phlogiston. In the above operation, ‘the oily principle’
referred to a substance contained in inflammable matters such as wood
or charcoal that had the strongest affinity with vitriolic acid. Common
or mineral sulphur was a ‘compound’ of this principle and vitriolic
acid, which accounted for its inflammability. In his earlier
experiments on metals with the burning glass, Geoffroy’s oily principle
was the ‘sulphur principle’ that regenerated metallic calces. Although
he could not isolate it in concrete form, he could attribute a distinct
chemical identity to this substance because it retained its peculiar
property of inflammability and its affinity for vitriolic acid
throughout various transformations. By identifying the ‘sulphur
principle’ in metallic chemistry with Stahl’s ‘phlogiston’ in the
chemistry of salts, Geoffroy broadened the operational terrain of the
‘sulphur principle’ or phlogiston. Furthermore, by including it in the Table
des rapports,
he provided phlogiston with a theoretical identity that would outlast
its operational identity. As the affinity table expanded and provided
more places for phlogiston in the chemistry of salts (Demachy 1781),
its place in chemical theory became more secure even as new questions
began to be raised about its operational identity as the principle of
inflammability, metallicity, and color. 4. The Thermometric Conception of FireIn the early decades of the eighteenth century, French academicians stabilized the ‘sulphur principle’ as ‘phlogiston’ by various operational/instrumental practices, a theoretical representation, and a corpuscular ontology. This complex of loosely coordinated identities attached to phlogiston faced a serious challenge when Herman Boerhaave (1668-1738), a popular teacher of chemistry and medicine, introduced a unified ontology of fire based on thermometric measurements. He utilized his scholarly training and inclination as a methodical reader to survey a broad range of literature and to construct a ‘true’ natural philosophy or experimental philosophy in the Boylean fashion. In trying to craft a didactic discourse of chemistry that would appeal to a more learned audience, he emphasized the intelligibility and the philosophical coherence of chemical theory. He sought to transform the ‘art’ of chemistry into an ‘academic science’ by employing the philosophical method of induction – to obtain ‘true knowledge’ through the ‘laborious’ process of proceeding from ‘particular experiments’ to ‘general rules’ (Boerhaave 1735, p. vii, ix) – which would allow him to present "the most perfect knowledge of chemistry" that utilized "all the labours of the chemists, which they call Processes". For this purpose, chemical ‘theory’ had to consist of the ‘general propositions’ deduced from "common undoubted chemical experiments" and common physical properties or the demonstrated ‘truths’ of physics, mechanics, hydrostatics and hydraulics (ibid., pp. 2-3). In other words, chemistry needed a shared instrumental basis with experimental physics to become a ‘science’. Chemistry’s ambiguous status as an art/science allowed Boerhaave to articulate a distinct epistemological use for its material practice. Chemistry "very justly deserves the name of an art" (ibid. p. 78), he argued, because it was a manipulative practice that utilized a set of ‘instruments’ to produce desired changes. Chemistry had six principal ‘instruments’ – fire, water, air, earth, the solvents, and the "common furniture of the Elaboratory" – which helped chemists to execute their ‘operations’. This instrumental practice indispensable to the ‘art’ of chemistry nevertheless made it particularly useful to natural philosophy because it created "new Phenomena" that "could have never been found out in the common order of nature". In contrast to the limited, accidental observations of Nature-as-is, chemistry cultivated instrumental or "physical Observations" that enriched the material foundation of natural philosophy. Boerhaave’s project of experimental philosophy is remarkably close to Lelas’ understanding of ‘experimentation’ – "to investigate nature by putting it on trial, by forcing it to behave in a new, induced way so that hidden properties not visible in its ‘natural’ spontaneous behaviour may be discovered" (Lelas 1993, p. 430). The first therefore, and indeed the principal part of this Science [Natural Philosophy], consists in collecting together all those Phaenomena of Bodies, which our senses are able to discover; and then reducing them into a Natural History. Now there are two different ways of coming at these Observations: The first, when we regard the appearances of things only as they happen indifferently to all in the common course of nature without any design in the human mind towards their production; and this is not of so great service to the Art, as in this case it is chance only that discovers to us certain properties, which happened to be produced at those particular times: The other, when we designedly apply different Bodies to one another that we are well acquainted with, purely with a view to observe the new Phaenomena that will arise from them. And this now, is of vastly more service to the Philosopher than the former: For, to mention no other reason, there are an infinite number of properties of Bodies, and those too very efficacious ones, that could never have been found out in the common order of nature, but then only discover themselves, when the Artist with his Fire in particular, comes to examine them, either jointly or separately, on purpose to know what will be the result. And, indeed, Chemistry is almost the only Art, that seems suited to cultivate this second, and most valuable method of making physical Observations. [Boerhaave 1735, pp. 50-51] The nature of fire commanded particular interest because it was the chief medium of chemical change and "the very instrument that Nature herself generally makes use of in the Operations it performs upon material beings" (ibid.). In other words, fire was the most pervasive and efficacious natural instrument chemists commanded to probe the inner secrets of nature. By employing this instrument of Nature, chemists could resolve compound bodies into their simple parts and combine them together again, thereby "discovering how it may exactly imitate the natural and common Phaenomena" (ibid.). It thus "truly explains, and exhibits to us the instruments by which nature so efficaciously operates; and thus pries into her most secret methods of working, and very often prudently directs and improves them to its own advantage" (ibid.). In the Traité du feu, therefore, Boerhaave advised ‘utmost caution’ in investigating the nature of fire so as not to create an erroneous foundation for natural philosophy. It is necessary, therefore, if we would keep clear of mistakes, to act with the utmost caution in our searches after a thing whose nature is so hidden and mysterious. And for this reason we must absolutely disengage our selves from all mere speculations, nor give into any precarious hypothesis, how plausibly soever contrived, unless we would run headlong into confusion, and uncertainties: For if at setting out we lay a wrong foundation, the errors that arise hence will diffuse themselves through all the branches of natural philosophy; since in all the production of natural effects, Fire, as I observed before, has always far the greatest share. [Ibid., p. 79] From the "most faithful, and diligent inquiries" of fire, Boerhaave could discern only one effect "which is always, and everywhere the same, perfectly inseparable from it, and constantly invariable in every kind of Object". He regarded the "expansion of Bodies by Fire […] effected in a Glass hermetically sealed" as a "true, certain, individual, and proper mark of Fire" (ibid., p. 85). In other words, Boerhaave identified the expansive property of heat, measured by the thermometer, as the true indicator of fire.[8] Although he mentioned five categories of phenomena attributed to fire (heat, light, color, expansion/rarefaction, and burning/melting), his concept of fire drew largely on the instrumental practice of thermometry, judging from the extensive list of experiments he included to illustrate the concept of fire: expansion by heat, contraction by cold, thermometric properties of various fluids, the notion of absolute cold, heat radiation, and so on. Boerhaave then sought to match this thermometric conception of heat with the chemical notion of fire through a careful reflection on the composition of bodies. In his scheme, fire was not the first ‘element’ that constituted all natural bodies, but the most important ‘instrument’ that produced desired alterations in bodies. Musing upon the expansion and eventual melting of a metal by heat, for example, he asked whether fire could "so attenuate and divide such a Body, that those corpuscles which are thus fluid, are in reality the very Elements of the Bodies, so long as they continue in this state?" Fire seemed to have the "surprising power" to divide bodies into their "ultimate parts" and to reach "the most intimate nature of Bodies" (ibid., pp. 88-89). He did not believe, however, that fire could destroy the ultimate particles or ‘Elements themselves’ or that the substance of other bodies could be converted into "the very substance of elementary Fire" in the combustion process. Partly on theological grounds, Boerhaave believed that "the very same Fire does always exist, in the same quantity, and without alteration." He thus disputed the long-standing experimental conclusion that fire increased the weight of metallic calces. Cold, or the absence of fire, contracted bodies to form solid mass and thus could create the "most intimate union" (ibid. pp. 91-92). In trying to present chemistry as a
respectable
academic science, while taking advantage of its material reach to
construct a ‘true’ natural philosophy, Boerhaave advocated a unified
ontology of nature based on universal instrumental practices. His
conception of fire was primarily based on the thermometric experiment
of expansion and contraction, which led to the conclusion that fire did
not contribute to the weight or to the substance of bodies. This made
it difficult for him to accept fire as a chemical substance. His
discourse on fire omitted phlogiston altogether. By staking an
elaborate claim embedded in a methodical discourse that fire and heat
must be the same ‘thing’, however, he forced later generations of
chemists and experimental physicists to scramble for a common
instrumental method to stabilize the ontology of fire. In other words,
he broke down the barrier between the disparate instrumental domains of
heat and fire to invent a material being that catered to both domains.
By doing so, he made heat an integral part of chemical constitution,
now envisaged as an equilibrium between attractive and repulsive
forces. It was this symmetric, discursive infiltration of heat and fire
into each other’s instrumental domain that Lavoisier had to sort out
later in his characterization of gases. 5. Textbook TraditionBoerhaave had a strong presence in the literary public sphere of the early Enlightenment (Ashworth 1977, Wellman 1992). Along with Stahl, he also had a formative influence on the French didactic discourse of chemistry during the second quarter of the eighteenth century when the transition in analytic method, the concomitant theoretical change, and the move away from the corpuscular philosophy all conspired to create a serious vacuum. At the operational level, chemists had to correlate the products of distillation with those of solution chemistry; at the theoretical level, they had to develop a new conception of chemical composition that was based on the chemistry of salts; at the philosophical level, Cartesian philosophy increasingly became a problematic language to rationalize chemical experience. The variegated operational/instrumental, theoretical, and philosophical identities of phlogiston did not cohere well in the changing instrumental and philosophical contexts. Guillaume-François Rouelle (1703-1770), a popular teacher at the Jardin du roi, mixed Boerhaave’s physicalist notion of fire based on thermometric practice and Stahl’s chemical notion of phlogiston as a substance without much regard for theoretical consistency. He modified Stahl’s hierarchical theory of composition to stipulate four elements or principles (phlogiston or fire, earth, water, and air) which formed higher levels of composition – mixts, compounds, and supercompounds. All four units – principles, mixts, compounds, and supercompounds – could form aggregates. Of the six instruments Boerhaave listed, he named fire, air, water, and earth as ‘natural’ instruments that also serve as elements. The other two, solvents and vessels, were ‘artificial’ instruments. Rouelle deviated significantly from both Boerhaave and Stahl, however, in identifying phlogiston as fire or an element-instrument that could either combine with other principles or serve as an instrument of chemical action by changing the physical state of bodies as in the rarefaction of air. The action of fire was limited to breaking up the molecules of aggregates, without being able to penetrate more intimate levels of composition. In explaining combustion phenomena, however, Rouelle identified the ‘inflammable principle’ or phlogiston as a mixt composed of fire and another principle. It existed in the oily part of the body, dissipated into the air upon combustion, and entered into new combinations afterwards. Too subtle to be isolated through chemical analysis, it united with various bodies with different strengths (Rouelle 1767). In short, Rouelle sought to piece together an ontology of fire that would function as phlogiston in chemical operations and as the matter of fire in other instrumental and discursive contexts. Pierre-Joseph Macquer (1718-1784), who wrote the most influential textbook of chemistry in the eighteenth century, also struggled to manage disparate meanings of phlogiston (Macquer 1749). Although he endorsed the four principles (fire, air, water, and earth), he defined them as the limits of chemical analysis rather than as the primordial, Aristotelian elements that made up all bodies. His notion of fire, built on Louis Lemery’s, distinguished between the ‘pure fire’ that acted on bodies to separate their parts and the ‘fixed fire’ that was called ‘inflammable principle, or sulphur principle, or phlogiston’. The principal property of pure, elementary fire was to penetrate and dilate bodies to establish a kind of equilibrium. It acted primarily to render a body fluid and could thus be called the principle of fluidity. Macquer thought that all bodies in nature were solid in nature, and that fire, essentially fluid, rendered them fluid. In other words, Macquer’s ‘pure fire’ incorporated the thermometric conception of fire. The combustion of inflammable matters indicated, however, that they contained the matter of fire as one of their principles. This ‘fixed fire’ differed from the elementary fire in that it did not communicate any heat or light to the body to which it was united. Nor did it render the body fluid. Most important, it could be transferred from one body to another, which made it plausible that it was a combination of the elementary fire with some other principle, which served as the base to form a kind of secondary principle. One could not isolate it in pure form, but the inflammability of a body would indicate that it contained phlogiston. Charcoal, an intimate combination of phlogiston and the earthly substance in vegetables and animals, facilitated the transmission of phlogiston to other substances. Macquer’s ‘fixed fire’ thus picked up the traditional notion of phlogiston. Rouelle’s and Macquer’s discussion of fire
and
phlogiston indicates that these prominent public teachers of chemistry
at the Jardin du roi sought to reconcile various meanings of the
sulphur principle or phlogiston that had developed in different
instrumental, theoretical, philosophical, and discursive contexts.
Taken individually, each of these meanings could be illustrated in
particular ‘instrumental’ contexts of chemical analysis, burning glass
analysis, and thermometric experiments. When mixed together textually,
however, these definitions of fire/phlogiston became a long list of
inchoate meanings that destabilized its identity. Most problematic was
the divergent characteristics of the (weightless) ‘matter of fire’
measured by the thermometer and (weighing) phlogiston as a chemical
substance. It took a new set of instrumental practices in pneumatic
chemistry to sort out the limits of representation regarding
fire/phlogiston. 6. ConclusionThe stability of phlogiston in eighteenth-century French chemistry was rooted in the operational identity of the ‘sulphur principle’ as an oily matter distilled from various substances and its theoretical identity as a component of salts. Geoffroy’s method of analysis and synthesis in probing the general composition of metals strengthened this operational/theoretical identity and allowed him to identify it with Stahl’s phlogiston. Embedded in the affinity table, phlogiston acquired a secure place in the evolving chemistry of salts, which provided a comprehensive framework of investigation for eighteenth-century French chemistry. As salts and affinity tables multiplied, the place of phlogiston in chemical theory became stronger. As a stable chemical substance with well-defined operational and theoretical identities, however, phlogiston also carried a speculative/philosophical identity as the matter of light or fire. Homberg’s efforts to correlate the products of chemical analysis with those of burning glass analysis through a corpuscular ontology had created a discursive identity for the ‘sulphur principle’ that allowed chemists to rationalize their esoteric practice for the lay audience. Although it took a leap of faith to link the instrumental practice of the burning glass to the ‘matter of light’, this speculative identity of the ‘sulphur principle’ allowed Louis Lemery to conceptualize the new ‘model system’ for chemical analysis – selective dissolution of metals in acids – in an intelligible manner. In working out the solvent action of fire in corpuscular language and thereby re-inscribing the operational identity of the ‘sulphur principle’ as the universal solvent, Lemery strengthened the discursive identity of the sulphur principle that coordinated its operational/theoretical identity with its corpuscular ontology. In other words, the complex ‘substance identity’ phlogiston acquired in the early decades of the eighteenth century was based on chemists’ manipulative capacity, theoretical framework, and ‘ontological attitude’. Determining what count as essential properties to define a chemical substance is a difficult problem (Schummer 2002) because this primary ‘object’ of chemical investigation is often a moving target that changes with the evolving laboratory practice, much like the ‘gene’ in genetics (Keller 2000). The ontological problem of what the scientific ‘object’ is cannot always be clearly demarcated from the epistemological problem of how one investigates it. The ‘object’ of chemical investigation often starts its career as an artifice constituted by the material culture of the laboratory and acquires a degree of naturalness only when it is embedded in an expanded classification of chemical/natural substances. Whether one considers chemical substances as natural or artificial depends critically on chemists’ ability to forge material connections between the chemical and the natural substances and to provide a comprehensive order of nature that includes them. Strange as it may seem to us, Homberg’s ‘true sulphur principle’ or the ‘matter of light’ had the ontological prestige of being a ‘natural’ body, while chemists’ ‘sulphur principle’ distilled from plants, animals, and minerals was regarded an artifice manufactured by fire. The burning glass supposedly revealed true nature, while distillation methods transformed the composition of natural bodies. Phlogiston could only be a ‘chemical’ substance from its operational/theoretical identity, but it acquired the status of a ‘natural’ body as the ‘matter of light’ or the ‘matter of fire’. Disciplinary hierarchy played a crucial role, then, in determining the naturalness of a chemical substance. A new instrumental practice can cause a serious rupture in the established conception and order of chemical/natural substances. J. Schummer has argued that the ‘chemical species identity’ based on chemical operations (assisted by physical measurements and theoretical constraints) underwent a fundamental transformation through the use of spectroscopy. By changing the role of molecular structure from a theoretical ‘property’ of a chemical substance to the very definition of a new ‘molecular species’, spectroscopy changed not only the material practice, but also the ‘ontological attitude’ of chemists.[9] Similarly, the thermometer introduced a serious ontological problem for eighteenth-century chemists. Although Louis Lemery’s differentiation between the ‘fixed fire and the ‘free fire’ opened a way to incorporate thermometric practice and conception of heat into chemical theory, Boerhaave brought to the chemists’ attention that such a facile comprise could not be tolerated at the ontological level. His effort to craft a singular ontology of material fire by fusing the chemical (burning glass) weighing fire and the thermometric weightless fire destabilized the ontological status of phlogiston as a natural substance. This discursive development, which served to distance phlogiston from its operational roots, created much confusion for the younger generation of chemists who learned chemistry as a public discourse rather than as the apothecary’s material practice. If Boerhaave’s philosophizing on chemical art made it more appealing to the literary public, it posed new problems for chemical teachers such as Rouelle and Macquer in reconciling their art with the philosophical discourse. By the 1760s, phlogiston had become primarily
a
textual entity in France, while the British chemists tried to re-invent
its operational identity through pneumatic chemistry (Eklund 1972, Basu
1992, Holmes 2000). Its discursive identity consisted of an ensemble of
its past identities without close association with the ongoing
investigation. Lavoisier targeted phlogiston because it hampered his
effort to rationalize chemistry as a logical discursive system with a
new set of ‘metric’ instruments (hydrometer, thermometer, gasometer,
and calorimeter) that would allow him to represent chemical action in
an algebraic form. He thus invented a new hypothetical substance,
caloric, that could absorb the philosophical identity of phlogiston as
the most subtle fluid, yet be measured by a ‘meter’, which would make
chemical action amenable to algebraic representation. In order to
understand Lavoisier’s ‘anti-phlogistic’ campaign, then, we need to go
beyond his rhetoric that phlogiston was an ‘imaginary’ substance. His
vision of rational chemistry was based on metric measurements and
expressed in algebraic terms. Phlogiston offered a unique resistance to
this vision and had to be eliminated (Kim 2003, pp. 279-389). AcknowledgementsI wish to thank members of the Triangle
History and Philosophy of Science Group for a lively discussion on the
first draft of this paper. Hasok Chang and an anonymous referee for Hyle
provided detailed comments that helped me to identify various problems
in getting the ideas across. Notes[1] I use the capitalized ‘Chemical Revolution’ here to refer to the theoretical revolution initiated by Lavoisier and his cohorts at the Paris Academy of Sciences. [2] Guerlac’s pioneering work (1961) was followed by many of his students’. For a bibliography on Lavoisier scholarship, see Bret 1995. [3] ‘Salts’ in the early eighteenth century referred to substances distilled from natural bodies that dissolved in water, which included acids, alkalis, and any combinations thereof, referred to as ‘middle salts’. [4] The ‘experimental system’ in Rheinberger 1997 is the ‘genuine working unit’ at each site of scientific research that differentially produces the object of investigation, which thereby captures the constantly mutating micro-dynamics of research. The ‘model system’ articulated in Creager 2002 refers to a communally shared experimental system such as the Tobacco Mosaic Virus that acquired a representative status to constitute a stable field of inquiry. [5] Russell McCormmach (2004) has shown that the object of natural philosophy was defined in the eighteenth century as ‘speculative truth’ because natural philosophers, despite their advocacy of accurate observations and ingenious experiments, would pursue a broad comprehension of nature including the conceptions of space, time, matter, and force. Their notion of ‘theory’ thus comprised conjectures, assertions, worldviews, hypotheses, natural laws, and causes with differing degrees of reliance on experimental practice. [6] Homberg stated that the old burning mirrors were nearly useless for chemical experiments because they used concave mirrors to focus the solar rays from below, which necessitated that the experimental material would have to be placed on an upside-down dish. The Tschirnhaus burning glass used convex glass to focus the solar rays directly on a regularly positioned dish. Over three feet in diameter, it provided a much more powerful furnace than any other available chemical furnace (Homberg 1702b). [7] Homberg could not use it for the analysis of common sulphur, for example, because of its high combustibility (Homberg 1703). For an excellent example of disputes regarding the credibility of the instrument, see Schaffer 1989. [8] Boerhaave worked closely with Fahrenheit (Van der Star 1983). [9] Schummer’s and
my position on
the epistemological/ontological function of instruments differ from
Rothbart and Slayden’s (1994) defense of ‘experimental realism’, which
limits the role of instruments largely to their epistemic value as the
analogs to natural systems. ReferencesAshworth, E.: 1977, Boerhaave’s Men at Leyden and After, Edinburgh: Edinburgh UP. Baird, D.: 2004, Thing Knowledge: A Philosophy of Scientific Instruments, Berkeley, CA: University of California Press. Basu, P.K.: 1992, ‘Similarities and Dissimilarities between Joseph Priestley’s and Antoine Lavoisier’s Chemical Beliefs’, Studies in History and Philosophy of Science, 23, 445-469. Boerhaave, H.: 1735, Elements of Chemistry, London: J. and J. Pemberton. Bret, P.: 1995, ‘Trois décennies d’études lavoisiennes’, Revue d’histoire des sciences, 48, 169-205. Conant, J.B.: 1967, The Overthrow of the Phlogiston Theory: The Chemical Revolution of 1775-1789, Cambridge, MA: Harvard UP. Creager, A. N. H.: 2002, Life of a Virus: Tobacco Mosaic Virus as an Experimental Model, 1930-1965, Chicago: University of Chicago Press. Davis, T. L.: 1928, ‘The Vicissitudes of Boerhaave’s Textbook of Chemistry’, Isis, 10, 33-46. Demachy, J.-F.: 1781, Recueil de Dissertations physico-chimiques, présentées à différentes académies, Paris: Nyon and Barrois. Donovan, A.: 1975, Philosophical Chemistry in the Scottish Enlightenment: The Doctrines and Discoveries of William Cullen and Joseph Black, Edinburgh, Edinburgh UP. Dorveaux, P.: 1931, ‘Etienne-François Geoffroy’, Revue d’histoire de la pharmacie, 2, 118-126. Duncan, A.M.: 1962, ‘Some Theoretical Aspects of Eighteenth-Century Tables of Affinity’, Annals of Science, 18, 177-196 & 217-232. Duncan, A. M.: 1996, Laws and Order in Eighteenth-Century Chemistry, Oxford: Clarendon. Eklund, J.: 1972, ‘Chemical Analyis and the Phlogiston Theory, 1738-1772: Prelude to Revolution’, Yale University, unpublished Ph.D. Dissertation. Fontenelle, B. le Bouvier de: 1699, ‘Preface to the History of the Academy of Sciences, from 1666-1699’, translated in: L.M. Marsak (ed.), The Achievement of Bernard le Bovier de Fontenelle, New York: Johnson Reprint, 1970, part IV, pp. 17-23. Fontenelle, B. le Bouvier de: 1715, ‘Éloge de Monsieur Homberg’, reprinted in: Éloges des academicians avec l’histoire de l’académie royale des sciences, La Haye: Isaac vander Kloot, 1740, vol. I, pp. 350-368. Fontenelle, B. le Bouvier de: 1731, ‘Éloge de M. Geoffroy’, Histoire de l’Académie royale des sciences, avec mémoires, Histoire [hereafter cited as Histoire], 93-100. Galison, P.: 1997, Image and Logic: A Material Culture of Microphysics, Chicago: University of Chicago Press. Geoffroy, E.F.: 1704, ‘Manière de recomposer le Soufre commun’, Histoire de l’Académie royale des sciences, avec mémoires, Mémoires [hereafter cited as Mémoires], 278-286. Geoffroy, E. F.: 1709, ‘Expériences sur les metaux, faites avec le Verre ardent du Palais Royal’, Mémoires, 162-176. Geoffroy, E.-F.: 1718, ‘Des différents Rapports observés en Chymie entre différentes substances’, Mémoires, 256-269. Geoffroy, E.-F.: 1720, ‘Eclaircissemens sur la Table insérée dans les Mémoires de 1718, concernant les rapports observés entre différentes substances’, Mémoires, 20-34. Gibbs, F.W.: 1958, ‘Boerhaave’s Chemical Writings’, Ambix, 6, 117-135. Golinski, J.: 1992, Science as Public Culture: Chemistry and Enlightenment in Britain, 1760-1820, Cambridge: Cambridge UP. Guerlac, H.: 1961, Lavoisier – The Crucial Year: The Background and Origin of His First Experiments on Combustion in 1772, Ithaca: Cornell UP. Harré, R.: 2003, ‘The Materiality of Instruments in a Metaphysics for Experiments’, in: H. Radder (ed.), The Philosophy of Scientific Experimentation, Pittsburgh, University of Pittsburgh Press, pp. 19-38. Holmes, F.L.: 1989, Eighteenth-Century Chemistry as an Investigative Enterprise, Berkeley: Office for History of Science and Technology. Holmes, F.L.: 1996, ‘The Communal Context for Etienne-François Geoffroy’s ‘Table des rapports’’, Science in Context, 9, 289-311. Holmes, F.L.: 2000, ‘The "Revolution in Chemistry and Physics": Overthrow of a Reigning Paradigm or Competition between Contemporary Research Programs?’, Isis, 91, 735-753. Homberg, W.: 1702a, ‘Essais de chimie’, Mémoires, 33-52. Homberg, W.: 1702b, ‘Observations faites par le moyen du Verre ardent’, Mémoires, 141-149. Homberg, W.: 1703, ‘Essay de l’analyse du Souffre commun’, Mémoires, 31-40. Homberg, W.: 1705a, ‘Sur le souffre Principe’, Procès-verbaux de l’Académie royal des sciences, 24, 125r-130v. Homberg, W.: 1705b, ‘Suite des Essays de chimie. Article Troisième. Du Souphre Principe’, Mémoires, 88-96. Homberg, W.: 1706, ‘Suite de l’article trois des Essais de chimie’, Mémoires, 260-272. Homberg, W.: 1708, ‘Mémoire touchant les Acides & les Alkalis’, Mémoires, 312-323. Ihde, D.: 1991, Instrumental Realism, Bloomington: Indiana UP. Keller, E.F.: 2000, The Century of the Gene, Cambridge, MA: Harvard UP. Kim, M.G.: 2000, ‘Chemical Analysis and the Domains of Reality: Wilhelm Homberg’s Essais de chimie, 1702-1709’, Studies in History and Philosophy of Science, 31, 37-69. Kim, M.G.: 2003, Affinity, That Elusive Dream: A Genealogy of the Chemical Revolution, MIT Press, Cambridge, MA. Kim, M.G.: 2006, ‘Experimental Systems and Theory Domains in pre-Lavoisian Chemistry’, 5th International Conference on History of Chemistry. Proceedings, SPQ, pp. 468-475. Klein, U.: 1995, ‘E.F. Geoffroy’s Table of different "Rapports" observed between different chemical substances – A Reinterpretation’, Ambix, 42, 251-287. Kroes, P.: 2003, ‘Physics, Experiments, and the Concept of Nature’, in: H. Radder (ed.), The Philosophy of Scientific Experimentation, Pittsburgh, University of Pittsburgh Press, pp. 68-86. Kuhn, T.S.: 1962, The Structure of Scientific Revolutions, Chicago: University of Chicago Press. Lafont, O.: 1991, ‘Personnalisation des rapports individu-puissance publique ou: Geoffroy et la famille Le Tellier’, Revue d’histoire de la pharmacie, 38, 15-23. Lelas, S.: 1993, ‘Science as Technology’, British Journal for the Philosophy of Science, 44, 423-442. Lemery, L.: 1709, ‘Conjectures & réflexions sur la matière du feu ou de la lumière’, Mémoires, 400-418. Lemery, L.: 1711, ‘Mémoire sur les précipitations Chimiques’, Mémoires, 56-79. Lemery, L.: 1712, ‘Conjectures sur les couleurs différentes des Précipités de Mercure’, Mémoires, 51-70. Macquer, P.-J.: 1749, Elémens de chymie – théorique, Paris: J.T. Hérissant. Margolis, H.: 1993, Paradigms & Barriers: How Habits of Mind Govern Scientific Beliefs, Chicago: University of Chicago Press. McCann, H.G.: 1978, Chemistry Transformed: The Paradigmatic Shift from Phlogiston to Oxygen, Norwood, NJ: Ablex. McCormmach, R.: 2004, Speculative Truth: Henry Cavendish, Natural Philosophy, and the Rise of Modern Theoretical Science, Oxford: Oxford UP. Musgrave, A.: 1976, ‘Why did oxygen supplant phlogiston? Research programmes in the Chemical Revolution’, in: C. Howson (ed.), Method and Appraisal in the Physical Sciences: The critical background to modern science, 1800-1905, Cambridge, Cambridge UP, pp. 181-209. Partington, J.R. & McKie, D.: 1937, ‘Historical Studies on the Phlogiston Theory – I. The Levity of Phlogiston’, Annals of Science, 2, 361-404. Partington, J.R. & McKie, D.: 1938, ‘Historical Studies on the Phlogiston Theory – II. The Negative Weight of Phlogiston’, Annals of Science, 3, 1-58. Perrin, C.E.: 1992, ‘The Chemical Revolution: Shifts in Guiding Assumptions’, in: A. Donovan et al. (eds.), Scrutinizing Science: Empirical Studies of Scientific Change, Baltimore: Johns Hopkins UP, pp. 105-124. Planchon, G.: 1898, ‘La dynastie des Geoffroy, apothicaires de Paris’, Journal de pharmacie et de chimie, 8, 289-293 and 337-345 Principe, L.M.: 2001, ‘Wilhelm Homberg: Chymical Corpuscularianism and Chrysopoeia in the Early Eighteenth Century’, in: C. Luthy et al. (eds.), Late Medieval and Early Modern Corpuscular Matter Theories, Leiden: Brill, pp. 535-556. Pyle, A.: 2000, ‘The Rationality of the Chemical Revolution’, in: R. Nola & H. Sankey (eds.), After Popper, Kuhn and Feyerabend, Dordrecht: Kluwer, pp. 99-124. Radder, H. (ed.): 2003, The Philosophy of Scientific Experimentation, Pittsburgh, University of Pittsburgh Press. Rheinberger, H.-J.: 1997, Toward a History of Epistemic Things, Stanford: Stanford UP. Rothbart, D. & Slayden, S.W.: 1994, ‘The Epistemology of a Spectrometer’, Philosophy of Science, 61, 25-38. Rouelle, G.-F.: 1767, ‘Cours de Chymie, recueilli des leçons de M. Rouelle’, Ms. 1202, Museum nationale d’histoire naturelle, Paris. Thagard, P.: 1990, ‘Conceptual Structure of the Chemical Revolution’, Philosophy of Science, 57, 183-209. Toulmin, S.E.: 1957, ‘Crucial Experiments: Priestley and Lavoisier’, Journal of the History of Ideas, 18, 205-220. Schaffer, S.: 1989, ‘Glass Works: Newton’s Prisms and the Uses of Experiment’, in: D. Gooding et al. (eds.), The Uses of Experiment, Cambridge: Cambridge UP, pp. 67-104. Schummer, J.: 2002, ‘The Impact of Instrumentation on Chemical Species Identity: From Chemical Substances to Molecular Species’, in: P.J.T. Morris (ed.), From Classical to Modern Chemistry: The Instrumental Revolution, London: The Royal Society of Chemistry, pp. 188-211. Senac, J.-B.: 1723, Nouveau Cours de chimie, suivant les principes de Newton & de Sthall, Paris: Jacques Vincent. Shelton, J.: 1995, ‘Seeing and Paradigms in the Chemical Revolution’, Philosophy of Science, 6, 129-141. Van der Star, P.: 1983, Fahrenheit’s Letters to Leibniz and Boerhaave, Leiden: Rodopi. Wellman, K.: 1992, La Mettrie: Medicine, Philosophy, and Enlightenment, Durham, NC: Duke UP. White, J.H.: 1932, The History of the Phlogiston Theory, London: Edward Arnold.
|
||||||||||||||||||||||||||||||||||||||||||||||||